



生物多样性 ›› 2012, Vol. 20 ›› Issue (4): 451-459. DOI: 10.3724/SP.J.1003.2012.09221 cstr: 32101.14.SP.J.1003.2012.09221
周蓉1, 李佳琦2, 李铀3, 刘迺发2,*( ), 房峰杰2, 施丽敏2, 王莹2
), 房峰杰2, 施丽敏2, 王莹2
收稿日期:2011-12-20
接受日期:2012-04-11
出版日期:2012-07-20
发布日期:2012-09-12
通讯作者:
刘迺发
作者简介:*E-mail: naifaliu@sohu.com基金资助:
Rong Zhou1, Jiaqi Li2, You Li3, Naifa Liu2,*( ), Fengjie Fang2, Limin Shi2, Ying Wang2
), Fengjie Fang2, Limin Shi2, Ying Wang2
Received:2011-12-20
Accepted:2012-04-11
Online:2012-07-20
Published:2012-09-12
Contact:
Naifa Liu
摘要:
大石鸡(Alectoris magna)是我国的特有鸟类, 其种群数量由于受到栖息地片断化和人类狩猎的影响而日益减少。本文以mtDNA的控制区部分序列(1,127 bp)和细胞色素b部分序列(807 bp)为分子标记, 研究了大石鸡分布区内12个种群234个样本的遗传变异。173个样本的控制区序列中共发现了34个多态位点, 定义了44种单倍型, 平均单倍型多样性和核苷酸多样性分别为0.916 ± 0.011和0.00449 ± 0.00242, 所有种群中单倍型多样性最高的是靖远和民和种群 (0.894 ± 0.063), 最低的是海原种群(0.476 ± 0.155)。230个样本的细胞色素b序列中共发现了13个多态位点, 定义了14种单倍型。平均单倍型多样性为0.738 ± 0.024, 平均核苷酸多样性为0.00216 ± 0.00009, 所有种群中单倍型多样性最高的是共和种群(0.763 ± 0.059), 最低的为德令哈种群(0.000 ± 0.000)。所有遗传多样性参数与样本量均无显著相关性。控制区和细胞色素b的单倍型邻接树均显示大石鸡在系统发生树上没有互为单系发生的种群存在, 因此应该将大石鸡作为一个独立的进化显著单元(ESU)来对待。AMOVA结果显示大部分遗传变异分布于种群内, 但种群间和群组间也存在显著的遗传变异, 所以应该把各个种群作为独立的管理单元(MU)来实现短期内的管理和保护。对德令哈、都兰、礼县、张家川、海原等遗传多样性较低的边缘种群应重点保护, 另外, 对共和、贵德和靖远3个遗传多样性较高的种群也应重点加以保护。
周蓉, 李佳琦, 李铀, 刘迺发, 房峰杰, 施丽敏, 王莹 (2012) 基于线粒体DNA的大石鸡种群遗传变异. 生物多样性, 20, 451-459. DOI: 10.3724/SP.J.1003.2012.09221.
Rong Zhou, Jiaqi Li, You Li, Naifa Liu, Fengjie Fang, Limin Shi, Ying Wang (2012) Genetic variation in rusty-necklaced partridge (Alectoris magna) detected by mitochondrial DNA. Biodiversity Science, 20, 451-459. DOI: 10.3724/SP.J.1003.2012.09221.
| 种群 Populations | 经纬度 Locality | 所属亚种 Subspecies | 样本量 Sample size | ||
|---|---|---|---|---|---|
| 控制区 D-loop | 细胞色素b CYTB | ||||
| 青海 Qinghai | |||||
| 德令哈 Delingha (DLH) | 37.22°N, 97.37°E | A. m. magna | 12 | 12 | |
| 乌兰 Wulan (WL) | 36.93°N, 98.48°E | A. m. magna | 18 | 23 | |
| 都兰 Dulan (DL) | 36.30°N, 98.10°E | A. m. magna | 25 | 34 | |
| 共和 Gonghe (GH) | 36.28°N, 100.62°E | A. m. lanzhouensis | 21 | 20 | |
| 贵德 Guide (GD) | 35.93°N, 101.48°E | A. m. lanzhouensis | 11 | 12 | |
| 互助 Huzhu (HZ) | 36.85°N, 102.15°E | A. m. lanzhouensis | 16 | 15 | |
| 民和 Minhe (MH) | 36.33°N, 102.80°E | A. m. lanzhouensis | 12 | 12 | |
| 甘肃 Gansu 靖远 Jingyuan (JY) | 36.34°N, 104.81°E | A. m. lanzhouensis | 12 | 40 | |
| 会宁 Huining (HN) | 35.91°N, 104.80°E | A. m. lanzhouensis | 8 | 16 | |
| 礼县 Lixian (LX) | 34.20°N, 105.13°E | A. m. lanzhouensis | 14 | 18 | |
| 张家川 Zhangjiachuan (ZJC) | 34.99°N, 106.21°E | A. m. lanzhouensis | 9 | 10 | |
| 宁夏 Ningxia 海原 Haiyuan (HY) | 36.57°N, 105.64°E | A. m. lanzhouensis | 15 | 18 | |
| 总计 Total | 173 | 230 | |||
表1 大石鸡12个种群的采样点信息
Table 1 Sampling information for 12 populations of Alectoris magna
| 种群 Populations | 经纬度 Locality | 所属亚种 Subspecies | 样本量 Sample size | ||
|---|---|---|---|---|---|
| 控制区 D-loop | 细胞色素b CYTB | ||||
| 青海 Qinghai | |||||
| 德令哈 Delingha (DLH) | 37.22°N, 97.37°E | A. m. magna | 12 | 12 | |
| 乌兰 Wulan (WL) | 36.93°N, 98.48°E | A. m. magna | 18 | 23 | |
| 都兰 Dulan (DL) | 36.30°N, 98.10°E | A. m. magna | 25 | 34 | |
| 共和 Gonghe (GH) | 36.28°N, 100.62°E | A. m. lanzhouensis | 21 | 20 | |
| 贵德 Guide (GD) | 35.93°N, 101.48°E | A. m. lanzhouensis | 11 | 12 | |
| 互助 Huzhu (HZ) | 36.85°N, 102.15°E | A. m. lanzhouensis | 16 | 15 | |
| 民和 Minhe (MH) | 36.33°N, 102.80°E | A. m. lanzhouensis | 12 | 12 | |
| 甘肃 Gansu 靖远 Jingyuan (JY) | 36.34°N, 104.81°E | A. m. lanzhouensis | 12 | 40 | |
| 会宁 Huining (HN) | 35.91°N, 104.80°E | A. m. lanzhouensis | 8 | 16 | |
| 礼县 Lixian (LX) | 34.20°N, 105.13°E | A. m. lanzhouensis | 14 | 18 | |
| 张家川 Zhangjiachuan (ZJC) | 34.99°N, 106.21°E | A. m. lanzhouensis | 9 | 10 | |
| 宁夏 Ningxia 海原 Haiyuan (HY) | 36.57°N, 105.64°E | A. m. lanzhouensis | 15 | 18 | |
| 总计 Total | 173 | 230 | |||
| 种群 Population | 控制区 D-loop | 细胞色素 b CYTB | |||
|---|---|---|---|---|---|
| h | π | h | π | ||
| 德令哈 DLH | 0.652 ± 0.133 | 0.00257 ± 0.00046 | 0.000 ± 0.000 | 0.00000 ± 0.00000 | |
| 乌兰 WL | 0.712 ± 0.074 | 0.00370 ± 0.00040 | 0.553 ± 0.080 | 0.00158 ± 0.00029 | |
| 都兰 DL | 0.663 ± 0.075 | 0.00386 ± 0.00046 | 0.508 ± 0.055 | 0.00124 ± 0.00014 | |
| 共和 GH | 0.800 ± 0.062 | 0.00342 ± 0.00035 | 0.763 ± 0.059 | 0.00202 ± 0.00022 | |
| 贵德 GD | 0.818 ± 0.119 | 0.00329 ± 0.00068 | 0.727 ± 0.113 | 0.00244 ± 0.00037 | |
| 互助 HZ | 0.700 ± 0.080 | 0.00303 ± 0.00068 | 0.467 ± 0.148 | 0.00090 ± 0.00032 | |
| 民和 MH | 0.894 ± 0.063 | 0.00330 ± 0.00055 | 0.591 ± 0.108 | 0.00207 ± 0.00037 | |
| 靖远 JY | 0.894 ± 0.063 | 0.00436 ± 0.00062 | 0.708 ± 0.062 | 0.00193 ± 0.00022 | |
| 会宁 HN | 0.750 ± 0.139 | 0.00219 ± 0.00105 | 0.508 ± 0.126 | 0.00154 ± 0.00036 | |
| 礼县 LX | 0.495 ± 0.151 | 0.00156 ± 0.00055 | 0.745 ± 0.079 | 0.00197 ± 0.00041 | |
| 张家川 ZJC | 0.694 ± 0.147 | 0.00197 ± 0.00064 | 0.644 ± 0.152 | 0.00173 ± 0.00054 | |
| 海原 HY | 0.476 ± 0.155 | 0.00208 ± 0.00087 | 0.471 ± 0.130 | 0.00116 ± 0.00045 | |
| 平均 Average | 0.916 ± 0.011 | 0.00449 ± 0.00242 | 0.738 ± 0.024 | 0.00216 ± 0.00009 | |
表2 大石鸡12个种群的单倍型多样性(h)和核苷酸多样性(π)
Table 2 Haplotype diversity (h) and nucleotide diversity (π) for 12 populations of Alectoris magna
| 种群 Population | 控制区 D-loop | 细胞色素 b CYTB | |||
|---|---|---|---|---|---|
| h | π | h | π | ||
| 德令哈 DLH | 0.652 ± 0.133 | 0.00257 ± 0.00046 | 0.000 ± 0.000 | 0.00000 ± 0.00000 | |
| 乌兰 WL | 0.712 ± 0.074 | 0.00370 ± 0.00040 | 0.553 ± 0.080 | 0.00158 ± 0.00029 | |
| 都兰 DL | 0.663 ± 0.075 | 0.00386 ± 0.00046 | 0.508 ± 0.055 | 0.00124 ± 0.00014 | |
| 共和 GH | 0.800 ± 0.062 | 0.00342 ± 0.00035 | 0.763 ± 0.059 | 0.00202 ± 0.00022 | |
| 贵德 GD | 0.818 ± 0.119 | 0.00329 ± 0.00068 | 0.727 ± 0.113 | 0.00244 ± 0.00037 | |
| 互助 HZ | 0.700 ± 0.080 | 0.00303 ± 0.00068 | 0.467 ± 0.148 | 0.00090 ± 0.00032 | |
| 民和 MH | 0.894 ± 0.063 | 0.00330 ± 0.00055 | 0.591 ± 0.108 | 0.00207 ± 0.00037 | |
| 靖远 JY | 0.894 ± 0.063 | 0.00436 ± 0.00062 | 0.708 ± 0.062 | 0.00193 ± 0.00022 | |
| 会宁 HN | 0.750 ± 0.139 | 0.00219 ± 0.00105 | 0.508 ± 0.126 | 0.00154 ± 0.00036 | |
| 礼县 LX | 0.495 ± 0.151 | 0.00156 ± 0.00055 | 0.745 ± 0.079 | 0.00197 ± 0.00041 | |
| 张家川 ZJC | 0.694 ± 0.147 | 0.00197 ± 0.00064 | 0.644 ± 0.152 | 0.00173 ± 0.00054 | |
| 海原 HY | 0.476 ± 0.155 | 0.00208 ± 0.00087 | 0.471 ± 0.130 | 0.00116 ± 0.00045 | |
| 平均 Average | 0.916 ± 0.011 | 0.00449 ± 0.00242 | 0.738 ± 0.024 | 0.00216 ± 0.00009 | |
| 变异来源 Source of variations | 变异组成 Variance components | 变异百分比 % of variation | P | 固定指数(99% IC) Fixation indices |
|---|---|---|---|---|
| D-loop | ||||
| 地理群组间 Among groups | 0.279 | 10.21 | 0.041 | FCT = 0.102 |
| 地理种群间 Among populations within groups | 0.534 | 19.54 | 0.000 | FSC = 0.218 |
| 种群内 Within populations | 1.921 | 70.25 | 0.000 | FST = 0.298 |
| CYTB | ||||
| 地理群组间 Among groups | 0.120 | 12.49 | 0.038 | FCT = 0.125 |
| 地理种群间 Among populations within groups | 0.205 | 21.33 | 0.000 | FSC = 0.244 |
| 种群内 Within populations | 0.635 | 66.18 | 0.000 | FST = 0.338 |
表3 控制区和细胞色素b部分序列分子变异分析(AMOVA)结果
Table 3 Analysis of molecular variance (AMOVA) of Alectoris magna based on partial sequences of D-loop and CYTB
| 变异来源 Source of variations | 变异组成 Variance components | 变异百分比 % of variation | P | 固定指数(99% IC) Fixation indices |
|---|---|---|---|---|
| D-loop | ||||
| 地理群组间 Among groups | 0.279 | 10.21 | 0.041 | FCT = 0.102 |
| 地理种群间 Among populations within groups | 0.534 | 19.54 | 0.000 | FSC = 0.218 |
| 种群内 Within populations | 1.921 | 70.25 | 0.000 | FST = 0.298 |
| CYTB | ||||
| 地理群组间 Among groups | 0.120 | 12.49 | 0.038 | FCT = 0.125 |
| 地理种群间 Among populations within groups | 0.205 | 21.33 | 0.000 | FSC = 0.244 |
| 种群内 Within populations | 0.635 | 66.18 | 0.000 | FST = 0.338 |
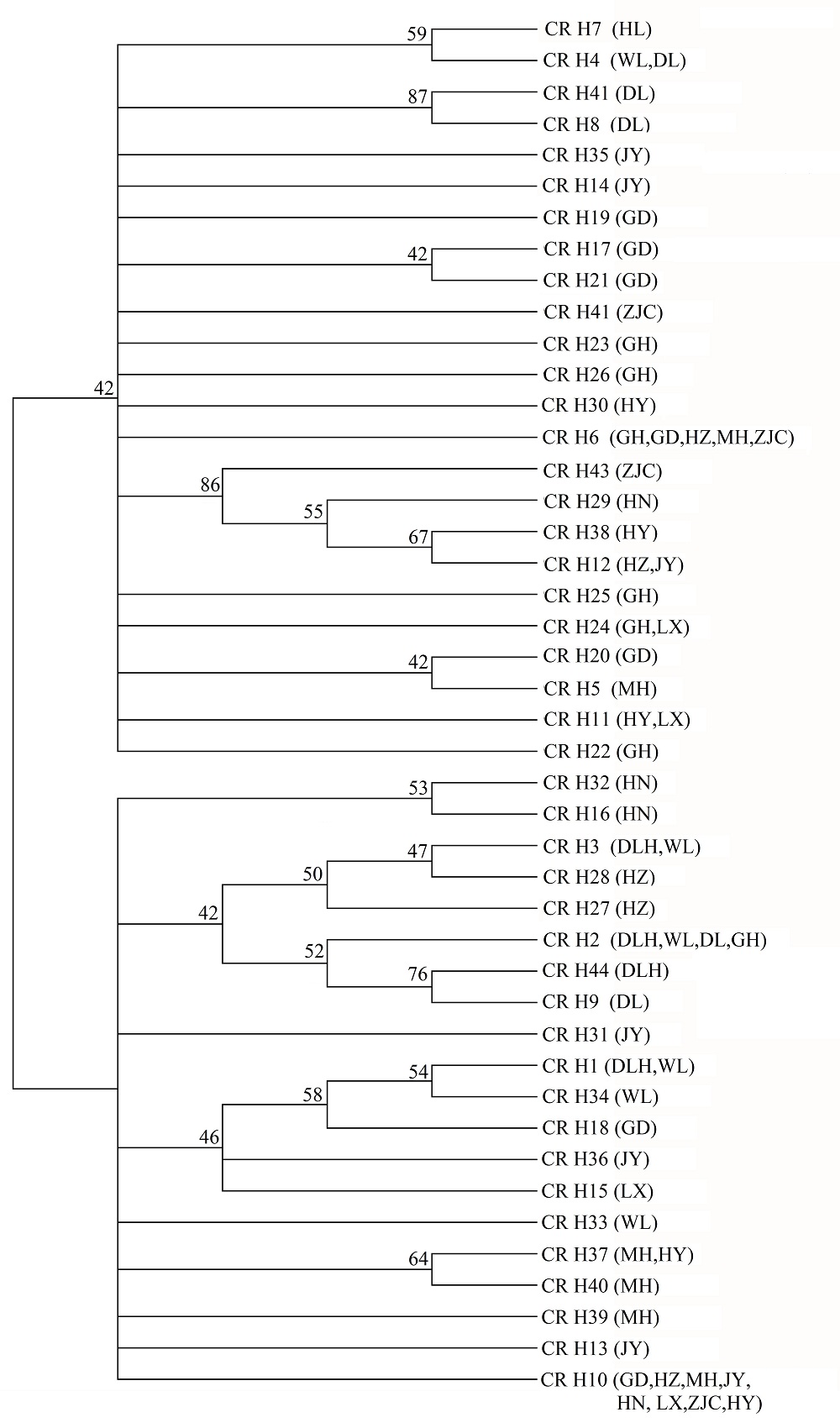
图1 用Tamura-Nei模型估计的遗传距离构建的线粒体DNA控制区单倍型的邻接树。数字表示经过1,000次重复抽样估算的每个节点的置信值。种群代号同表1。
Fig. 1 Neighbour-joining tree of Tamura-Nei’s distance between 44 mtDNA D-loop haplotypes. Numbers indicate the percentage con?dence level of each node estimated by 1,000 bootstrap samplings of the data. Population codes see Table 1.
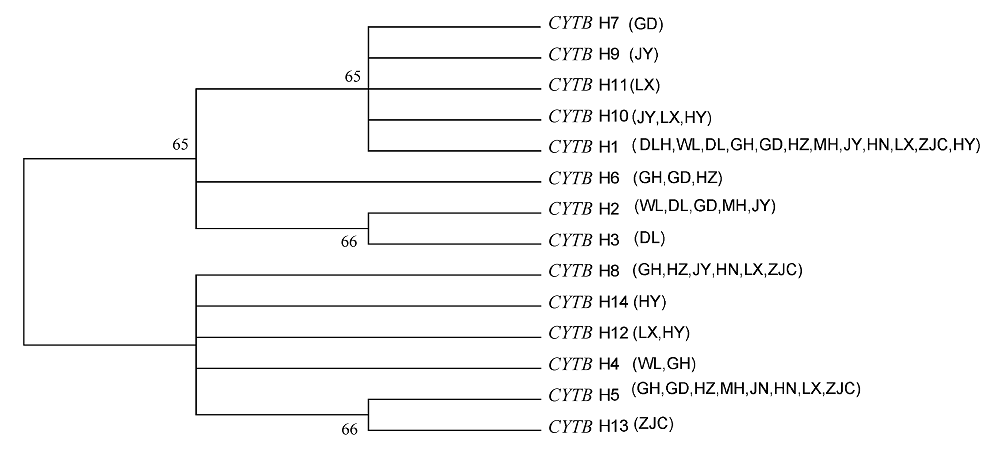
图2 用Tamura-Nei模型估计的遗传距离, 构建线粒体DNA细胞色素b单倍型的邻接树, 数字表示经过1,000次重复抽样估算的每个节点的置信值。种群代号同表1。
Fig. 2 Neighbour-joining tree of Tamura-Nei’s distance between 14 mtDNA CYTB haplotypes. Numbers indicate the percentage con?dence level of each node estimated by 1,000 bootstrap samplings of the data. Population codes see Table 1.
| [1] | Avise JC (1994) Molecular Markers, Natural History and Evolution. Chapman & Hall, New York. |
| [2] | Avise JC, Hamrick JL (1996) Conservation Genetics: Case Histories from Nature. Chapman & Hall, New York. |
| [3] |
Barratt EM, Gurnell J, Malarky G, Deaville R, Bruford MW (1999) Genetic structure of fragmented populations of red squirrel (Sciurus vulgaris) in the UK. Molecular Ecology, 8, S55-S63.
URL PMID |
| [4] | Beebee TJC, Rowe G (translated by Zhang JL (张军丽), Liao B (廖斌), Wang SL (王胜龙) (2009) An Introduction to Molecular Ecology (分子生态学) , pp. 183-184. Sun Yat-sen University Press, Guangzhou. (in Chinese) |
| [5] | Chen Q, Chang C, Liu NF, Randi E, Lucchini V (1999) Mitochondrial DNA introgression between two parapatric species of Alectoris. Acta Zoologica Sinica, 45, 456-463. |
| [6] |
Excoffier L, Laval G, Schneider S (2005) Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evolutionary Bioinformatics Online, 1, 47-50.
URL PMID |
| [7] | Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics, 131, 479-491. |
| [8] |
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 39, 783-791.
DOI URL PMID |
| [9] |
García-Ramos G, Kirkpatrick M (1997) Genetic models of adaptation and gene flow in peripheral populations. Evolution, 51, 21-28.
URL PMID |
| [10] |
Garner TWJ, Pearman PB, Angelone S (2004) Genetic diversity across a vertebrate species’ range: a test of the central- peripheral hypothesis. Molecular Ecology, 13, 1047-1053.
DOI URL PMID |
| [11] | Groom MJ, Meffe GK, Carroll CR (2006) Principles of Conservation Biology. Sinauer Associates, Sunderland, Massachusetts, USA. |
| [12] |
Hoffmann AA, Blows MW (1994) Species borders: ecological and evolutionary perspectives. Trends in Ecology and Evolution, 9, 223-227.
DOI URL PMID |
| [13] | Hou P (侯鹏), Wei M (卫明), Zhang LX (张立勋), Liu NF (刘迺发) (2002) Genetic structure of edge population in Prze- walski’s rock partridge (Alectoris magna). Acta Zoologica Sinica (动物学报), 48, 333-338. (in Chinese with English abstract) |
| [14] |
Houlden BA, Costello BH, Sharkey D, Fowler EV, Melzer A, Ellis W, Carrick F, Baverstock PR, Elphinstone MS (1999) Phylogeographic differentiation in the mitochondrial control region in the koala, Phascolarctos cinereus (Goldfuss 1817). Molecular Ecology, 8, 999-1011.
DOI URL PMID |
| [15] | Huang ZH, Liu NF, Chen YK, Xiao YA (2009a) Genetic diver- sity in peripheral and central populations of rusty-necklaced partridge (Alectoris magna) based on mitochondrial and microsatellite DNA. Acta Zoologica Academiae Scientiarum Hungaricae, 55, 187-197. |
| [16] | Huang ZH, Liu NF, Luo SX, Long J, Xiao YA (2007) Ecological genetics of rusty-necklaced partridge (Alectoris magna): environmental factors and population genetic variability correlations. Korean Journal of Genetics, 29, 115-120. |
| [17] | Huang ZH, Yang ZS, Zhang J, Liu NF (2009b) Introgressive hybridization and population genetic diversity between rusty-necklaced partridge and chukar partridge in northwe- stern China. Belgian Journal of Zoology, 139, 15-21. |
| [18] | Karron JD (1987) A comparison of levels of genetic polymorphism and self-compatibility in geographically restricted and widespread plant congeners. Evolutionary Ecology, 1, 47-58. |
| [19] |
Kimura M (1968) Evolutionary rate at the molecular level. Nature, 217, 624-626.
DOI URL PMID |
| [20] | Lesica P, Allendorf FW (1995) When are peripheral popula- tions valuable for conservation? Conservation Biology, 9, 753-760. |
| [21] | Librado P, Rozas J (2009) DnaSP V5: a software for compre- hensive analysis of DNA polymorphism data. Bioin- formatics, 25, 1451-1452. |
| [22] | Liu NF (刘迺发) (1984) On the taxonomic status of Alectoris magna. Acta Zootaxonomica Sinica (动物分类学报), 9, 212-218. (in Chinese with English abstract) |
| [23] | Liu NF (1992) Ecology of Przewalski’s rock partridge (Alectoris magna). Gibier Faune Sauvage, 9, 605-615. |
| [24] | Liu NF (刘迺发), Huang ZH (黄族豪), Wen LY (文陇英) (2004) Subspecies divergence of Przewalski’s rock partridge (Alectoris magna) description of a new subspecies. Acta Zootaxonomica Sinica (动物分类学报), 29, 600-605. (in Chinese with English abstract) |
| [25] | Liu NF (刘迺发), Wang XT (王香亭), Luo WY (罗文英) Chang C (常城) (1988) Avifauna study of the Liupan Mountain in Ningxia. Journal of Lanzhou University (Natural Science Edition) (兰州大学学报 (自然科学版)), 24, 65-75. (in Chinese with English abstract) |
| [26] | Liu NF (刘迺发), Wen LY (文陇英), Huang ZH (黄族豪), Hou P (侯鹏) (2006) Introgressive hybridization between Alectoris magna and A. chukar in the Liupan Mountain Region. Acta Zoologica Sinica (动物学报), 52, 153-159. (in English with Chinese abstract) |
| [27] | Merrell DJ (1981) Ecological Genetics. University of Minnesota Press, Longman, London. |
| [28] |
Moritz C (1994) Defining ‘evolutionarily significant units’ for conservation. Trends in Ecology and Evolution, 9, 373-375.
DOI URL PMID |
| [29] | Moritz C, Dowling TE, Brown WM (1987) Evolution of animal mitochondrial DNA: relevance for population biology and systematics. Annual Review of Ecology and Systematics, 18, 269-292. |
| [30] |
Qu JY, Liu NF, Bao XK, Wang XL (2009) Phylogeography of the ring-necked pheasant (Phasianus colchicus) in China. Molecular Phylogenetics and Evolution, 52, 125-132.
DOI URL PMID |
| [31] | Qu YH, Lei FM (2009) Comparative phylogeography of two endemic birds of the Tibetan Plateau, the white-rumped snow finch (Onychostruthus taczanowskii) and the Hume’s ground tit (Pseudopodoces humilis). Molecular Phyloge- netics and Evolution, 51, 312-326. |
| [32] |
Qu YF, Lei FM, Zhang RY, Lu X (2010) Comparative phylogeography of five avian species: implications for Pleistocene evolutionary history in the Qinghai-Tibetan Plateau. Molecular Ecology, 19, 338-351.
DOI URL PMID |
| [33] |
Randi E, Lucchini V (1998) Organization and evolution of the mitochondrial DNA control region in the avian genus Alectoris. Journal of Molecular Evolution, 47, 449-462.
DOI URL PMID |
| [34] |
Rowe G, Beebee TJC (2003) Population on the verge of a mutational meltdown? Fitness costs of genetic load for an amphibian in the wild. Evolution, 57, 177-181.
DOI URL PMID |
| [35] | Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, New York. |
| [36] | Soule M (1973) The epistasis cycle: a theory of marginal populations. Annual Review of Ecology and Systematics, 4, 165-187. |
| [37] |
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 25, 4876-4882.
DOI URL PMID |
| [38] | Wang YQ (汪永庆), Zhang ZB (张知彬), Xu LX (徐来祥) (2002) The genetic diversity of central and peripheral populations of ratlike Hmster (Cricetulus triton). Chinese Science Bulletin (科学通报), 47, 201-206. (in Chinese) |
| [39] | Wei M (卫明), Hou P (侯鹏), Huang ZH (黄族豪), Liu NF (刘迺发) (2002) Effects of environmental factors on the population genetic structure in Alectoris magna. Acta Ecologica Sinica (生态学报), 22, 528-534. (in Chinese with English abstract) |
| [40] | Wen LY (文陇英), Zhang LX (张立勋), Liu NF (刘迺发) (2005) Phylogenetic relationship of Perdix dauuricae inferred from mitochondrial cytochrome b gene. Zoological Research (动物学研究), 26, 69-75. (in Chinese with English abstract) |
| [41] | Yang ZS (杨志松), Liu NF (刘迺发) (2009) Introgressive hybridization between two partridges based on mitochon- drial cytochrome b (Cytb) gene. Journal of Chongqing Normal University (Natural Science Edition) (重庆师范大学学报 (自然科学版)), 26, 1-6. (in Chinese with English abstract) |
| [1] | 洪德元. 分类学中的方法论小叙[J]. 生物多样性, 2025, 33(2): 24541-. |
| [2] | 王嘉陈, 徐汤俊, 许唯, 张高季, 尤艺瑾, 阮宏华, 刘宏毅. 城市景观格局对大蚰蜒种群遗传结构的影响[J]. 生物多样性, 2025, 33(1): 24251-. |
| [3] | 蒋陈焜, 郁文彬, 饶广远, 黎怀成, Julien B. Bachelier, Hartmut H. Hilger, Theodor C. H. Cole. 植物系统发生海报——以演化视角介绍植物多样性的科教资料项目[J]. 生物多样性, 2024, 32(11): 24210-. |
| [4] | 李庆多, 栗冬梅. 全球蝙蝠巴尔通体流行状况分析[J]. 生物多样性, 2023, 31(9): 23166-. |
| [5] | 冯晨, 张洁, 黄宏文. 统筹植物就地保护与迁地保护的解决方案: 植物并地保护(parallel situ conservation)[J]. 生物多样性, 2023, 31(9): 23184-. |
| [6] | 齐海玲, 樊鹏振, 王跃华, 刘杰. 中国北方六省区胡桃的遗传多样性和群体结构[J]. 生物多样性, 2023, 31(8): 23120-. |
| [7] | 熊飞, 刘红艳, 翟东东, 段辛斌, 田辉伍, 陈大庆. 基于基因组重测序的长江上游瓦氏黄颡鱼群体遗传结构[J]. 生物多样性, 2023, 31(4): 22391-. |
| [8] | 蒲佳佳, 杨平俊, 戴洋, 陶可欣, 高磊, 杜予州, 曹俊, 俞晓平, 杨倩倩. 长江下游外来生物福寿螺的种类及其种群遗传结构[J]. 生物多样性, 2023, 31(3): 22346-. |
| [9] | 何艺玥, 刘玉莹, 张富斌, 秦强, 曾燏, 吕振宇, 杨坤. 梯级水利工程背景下的嘉陵江干流蛇鮈群体遗传多样性和遗传结构[J]. 生物多样性, 2023, 31(11): 23160-. |
| [10] | 孙维悦, 舒江平, 顾钰峰, 莫日根高娃, 杜夏瑾, 刘保东, 严岳鸿. 基于保护基因组学揭示荷叶铁线蕨的濒危机制[J]. 生物多样性, 2022, 30(7): 21508-. |
| [11] | 陶克涛, 白东义, 图格琴, 赵若阳, 安塔娜, 铁木齐尔·阿尔腾齐米克, 宝音德力格尔, 哈斯, 芒来, 韩海格. 基于基因组SNPs对东亚家马不同群体遗传多样性的评估[J]. 生物多样性, 2022, 30(5): 21031-. |
| [12] | 崔静, 徐明芳, 章群, 李瑶, 曾晓舒, 李莎. 基于3种线粒体标记探讨中日沿海角木叶鲽遗传多样性差异[J]. 生物多样性, 2022, 30(5): 21485-. |
| [13] | 孙军, 宋煜尧, 施义锋, 翟键, 燕文卓. 近十年中国海洋生物多样性研究进展[J]. 生物多样性, 2022, 30(10): 22526-. |
| [14] | 栗冬梅, 杨卫红, 李庆多, 韩茜, 宋秀平, 潘虹, 冯云. 巴尔通体在滇西南蝙蝠中高度流行并具有丰富的遗传变异特征[J]. 生物多样性, 2021, 29(9): 1245-1255. |
| [15] | 陆奇丰, 黄至欢, 骆文华. 极小种群濒危植物广西火桐、丹霞梧桐的叶绿体基因组特征[J]. 生物多样性, 2021, 29(5): 586-595. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
备案号:京ICP备16067583号-7
Copyright © 2026 版权所有 《生物多样性》编辑部
地址: 北京香山南辛村20号, 邮编:100093
电话: 010-62836137, 62836665 E-mail: biodiversity@ibcas.ac.cn
![]()