



Biodiv Sci ›› 2011, Vol. 19 ›› Issue (3): 369-376. DOI: 10.3724/SP.J.1003.2011.08193 cstr: 32101.14.SP.J.1003.2011.08193
Special Issue: 土壤生物与土壤健康
Previous Articles Next Articles
Yunfu Gu1, Xiaoping Zhang1,*( ), Shihua Tu2, Kristina Lindström3
), Shihua Tu2, Kristina Lindström3
Received:2010-08-06
Accepted:2011-02-05
Online:2011-05-20
Published:2013-12-10
Contact:
Xiaoping Zhang
Yunfu Gu, Xiaoping Zhang, Shihua Tu, Kristina Lindström. Effect of long-term fertilization on archaeal community structure in calcareous purplish paddy soil[J]. Biodiv Sci, 2011, 19(3): 369-376.
| 施肥方式 Fertilization treatments | ||||||||
|---|---|---|---|---|---|---|---|---|
| M | NM | NPM | NPKM | CK | N | NP | NPK | |
| 水稻收获后 After rice cultivation | 4.043 | 3.993 | 4.987 | 4.057 | 3.969 | 4.990 | 3.942 | 3.753 |
| 小麦收获后 After wheat cultivation | 4.855 | 4.887 | 4.232 | 4.209 | 4.173 | 4.783 | 4.075 | 4.787 |
Table 1 Water content of air-dried top soil under different fertilization treatments after rice/wheat cultivation (%)
| 施肥方式 Fertilization treatments | ||||||||
|---|---|---|---|---|---|---|---|---|
| M | NM | NPM | NPKM | CK | N | NP | NPK | |
| 水稻收获后 After rice cultivation | 4.043 | 3.993 | 4.987 | 4.057 | 3.969 | 4.990 | 3.942 | 3.753 |
| 小麦收获后 After wheat cultivation | 4.855 | 4.887 | 4.232 | 4.209 | 4.173 | 4.783 | 4.075 | 4.787 |
| 引物 Primer | 16S rDNA目标区(碱基数) 16S rDNA target (base number) | 引物序列 Primer sequence |
|---|---|---|
| PRA46F | Archaea (46-60) | 5′- C/TTAAGCCATGCG/AAGT-3′ |
| PREA1100R | Archaea (1100-1117) | 5′- T/CGGGTCTCGCTCGTTG/ACC-3′ |
| PARCH340F-GC | Archaea V3 region (340-358) | 5′- CCTACGGGGC/TGCAG/CCAG-3′ |
| PARCH519R | Archaea V3 region (519-534) | 5′- TTACCGCGGCG/TGCTG-3′ |
Table 2 Specific amplification primers for the archaeal 16S rDNA in DGGE experiment (adopted from ?vre?s et al., 1997)
| 引物 Primer | 16S rDNA目标区(碱基数) 16S rDNA target (base number) | 引物序列 Primer sequence |
|---|---|---|
| PRA46F | Archaea (46-60) | 5′- C/TTAAGCCATGCG/AAGT-3′ |
| PREA1100R | Archaea (1100-1117) | 5′- T/CGGGTCTCGCTCGTTG/ACC-3′ |
| PARCH340F-GC | Archaea V3 region (340-358) | 5′- CCTACGGGGC/TGCAG/CCAG-3′ |
| PARCH519R | Archaea V3 region (519-534) | 5′- TTACCGCGGCG/TGCTG-3′ |
| 引物 Primers | 16S rDNA目标区(碱基数) 16S rDNA target (base number) | 引物序列 Primer sequence | 参考文献 References |
|---|---|---|---|
| Ar4F | Archaea (8-25) | 5′- TCY GGT TGA TCC TGC CRG-3′ | |
| Ar958R | Archaea (958-967) | 5′- YCC GGC GTT GAV TCC AAT T-3′ | |
| Ar9R | Archaea (906-927) | 5′-CCC GCC AAT TCC TTT AAG TTT C-3′ | |
| Ar3F | Archaea (7-26) | 5′- TTC CGG TTG ATC CTG CCG GA-3′ |
Table 3 Specific amplification primers for the archaeal 16S rDNA in RFLP experiment
| 引物 Primers | 16S rDNA目标区(碱基数) 16S rDNA target (base number) | 引物序列 Primer sequence | 参考文献 References |
|---|---|---|---|
| Ar4F | Archaea (8-25) | 5′- TCY GGT TGA TCC TGC CRG-3′ | |
| Ar958R | Archaea (958-967) | 5′- YCC GGC GTT GAV TCC AAT T-3′ | |
| Ar9R | Archaea (906-927) | 5′-CCC GCC AAT TCC TTT AAG TTT C-3′ | |
| Ar3F | Archaea (7-26) | 5′- TTC CGG TTG ATC CTG CCG GA-3′ |
| 施肥处理 Fertilization treatments | Shannon多样性指数 Shannon's diversity index (H) | 丰富度 Richness (S) | 均匀度 Evenness (EH) | |||||
|---|---|---|---|---|---|---|---|---|
| 水稻 Rice | 小麦 Wheat | 水稻 Rice | 小麦 Wheat | 水稻 Rice | 小麦 Wheat | |||
| M | 3.08±0.13a | 3.13±0.14a | 21±2.00a | 20±2.00a | 1.016±0.00a | 1.014±0.00a | ||
| NM | 2.94±0.09a | 2.99±0.07ab | 18±1.15b | 19±1.53ab | 1.017±0.00a | 1.018±0.01a | ||
| NPM | 3.04±0.09a | 3.09±0.09a | 20±1.53a | 21±1.53a | 1.014±0.05a | 1.015±0.00a | ||
| NPKM | 2.99±0.09a | 2.99±0.03ab | 19±0.58b | 19±1.53ab | 1.017±0.05a | 1.017±0.01a | ||
| CK | 3.09±0.07a | 3.04±0.07ab | 21±1.16a | 20±1.15a | 1.015±0.05a | 1.015±0.00a | ||
| N | 2.99±0.09a | 3.08±0.03ab | 19±0.06b | 21±1.53a | 1.014±0.00a | 1.017±0.00a | ||
| NP | 2.89±0.09b | 2.89±0.07b | 17±1.15b | 17±1.53b | 1.019±0.00a | 1.020±0.01a | ||
| NPK | 3.13±0.16a | 3.04±0.11ab | 22±2.08a | 20±3.00a | 1.013±0.03a | 1.014±0.00a | ||
Table 4 Archaea diversity as indicated by the number of DNA bands, Shannon diversity index (H), richness (S), and evenness (EH)
| 施肥处理 Fertilization treatments | Shannon多样性指数 Shannon's diversity index (H) | 丰富度 Richness (S) | 均匀度 Evenness (EH) | |||||
|---|---|---|---|---|---|---|---|---|
| 水稻 Rice | 小麦 Wheat | 水稻 Rice | 小麦 Wheat | 水稻 Rice | 小麦 Wheat | |||
| M | 3.08±0.13a | 3.13±0.14a | 21±2.00a | 20±2.00a | 1.016±0.00a | 1.014±0.00a | ||
| NM | 2.94±0.09a | 2.99±0.07ab | 18±1.15b | 19±1.53ab | 1.017±0.00a | 1.018±0.01a | ||
| NPM | 3.04±0.09a | 3.09±0.09a | 20±1.53a | 21±1.53a | 1.014±0.05a | 1.015±0.00a | ||
| NPKM | 2.99±0.09a | 2.99±0.03ab | 19±0.58b | 19±1.53ab | 1.017±0.05a | 1.017±0.01a | ||
| CK | 3.09±0.07a | 3.04±0.07ab | 21±1.16a | 20±1.15a | 1.015±0.05a | 1.015±0.00a | ||
| N | 2.99±0.09a | 3.08±0.03ab | 19±0.06b | 21±1.53a | 1.014±0.00a | 1.017±0.00a | ||
| NP | 2.89±0.09b | 2.89±0.07b | 17±1.15b | 17±1.53b | 1.019±0.00a | 1.020±0.01a | ||
| NPK | 3.13±0.16a | 3.04±0.11ab | 22±2.08a | 20±3.00a | 1.013±0.03a | 1.014±0.00a | ||
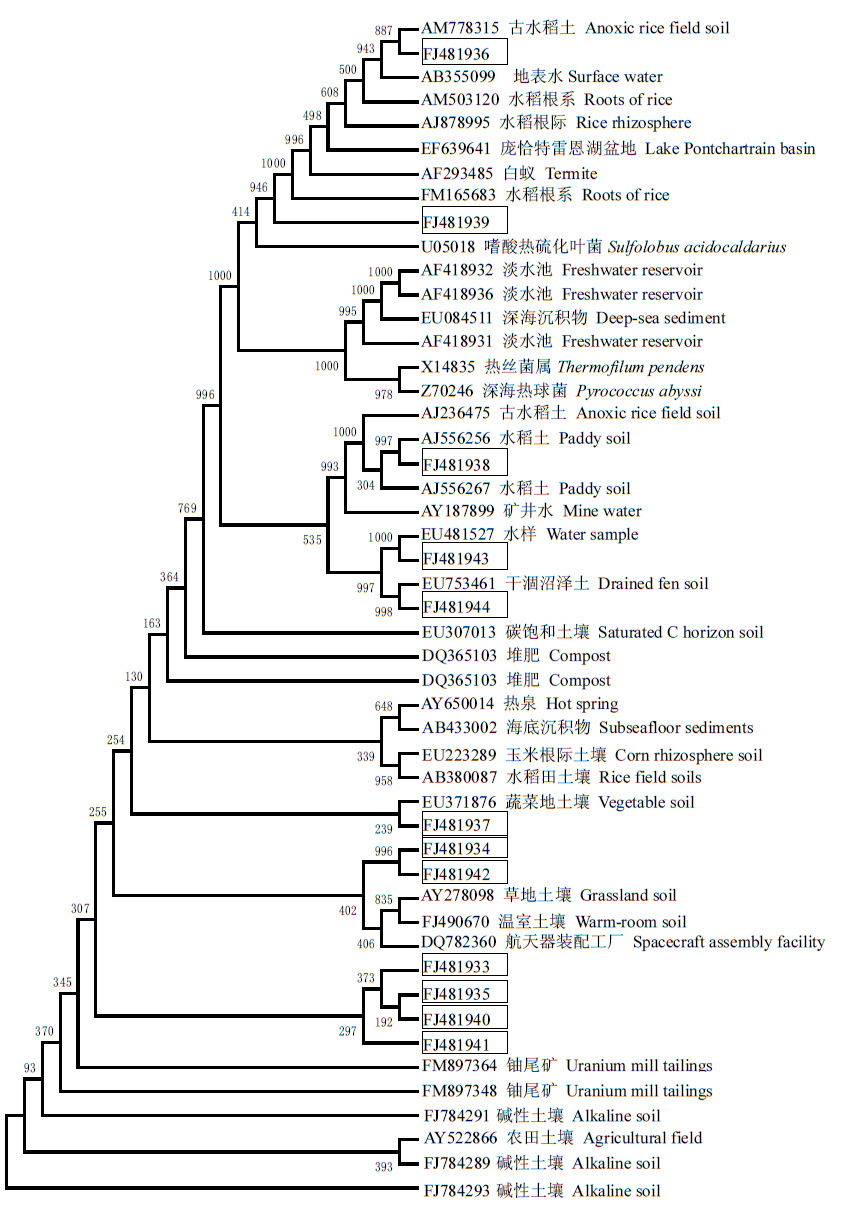
Fig. 3 Phylogenetic tree based on archaeal 16S rDNA library and some other known archaeal 16S rDNA sequences. The sequences in squares were isolated in this study.
| [1] |
Bastida F, Zsolnay A, Hernandez T, Garcia C (2008) Past, present and future of soil quality indices: a biological perspective. Geoderma, 147, 159-171.
DOI URL |
| [2] | Brock TD (1987) The study of microorganisms in situ: progress and problems. Symposia of the Society for General Microbiology, 41, 1-17. |
| [3] |
Chen X, Zhang LM, Shen JP, Xu ZH, He JZ (2010) Soil type determines the abundance and community structure of ammonia-oxidizing bacteria and archaea in flooded paddy soils. Journal of Soils and Sediments, 10, 1510-1516.
DOI URL |
| [4] |
Chen XP, Zhu YG, Xia Y, Shen JP, He JZ (2008) Ammonia- oxidizing archaea: important players in paddy rhizosphere soil? Environmental Microbiology, 10, 1978-1987.
URL PMID |
| [5] | Conrad R, Klose M (2005) Effect of potassium phosphate fertilization on production and emission of methane and its 13C-stable isotope composition in rice microcosms. Soil Biology and Biochemistry, 37, 2099-2108. |
| [6] | Delong EF, Pace NR (2001) Environmental diversity of bacteria and archaea. BMC System Biology, 50, 470-478. |
| [7] | Di HJ, Cameron KC, Shen JP, Winefield CS, O’Callaghan M, Bowatte S, He JZ (2009) Nitrification driven by bacteria and not archaea in nitrogen-rich grassland soils. Nature Geoscience, 2, 621-624. |
| [8] |
Fournier GP, Dick AA, Williams D, Peter GJ (2011) Evolution of the archaea: emerging views on origins and phylogeny. Research in Microbiology, 162, 92-98.
URL PMID |
| [9] | Garcia JL, Patel BKC, Ollivier B (2000) Taxonomic, phylogenetic, and ecological diversity of methanogenic archaea. Anaerobe, 6, 205-226. |
| [10] | Gu YF, Zhang XP, Tu SH, Vestberg M, Lindström K (2009) Soil microbial biomass, crop yields, and bacterial community structure as affected by long-term fertilization treatments under wheat-rice cropping. European Journal of Soil Biology, 45, 239-246. |
| [11] |
He JZ, Shen JP, Zhang LM, Zhu YG, Zheng YM, Xu MG, Di HJ (2008) Quantitative analyses of the abundance and composition of ammonia-oxidizing bacteria and ammonia-oxidizing archaea of a Chinese upland red soil under long-term fertilization practices. Environmental Microbiology, 9, 2364-2374.
DOI URL PMID |
| [12] |
Hershberger KL, Barns SM, Reysenbach AL, Dawson SC, Pace NR (1996) Wide diversity of Crenarchaeota. Nature, 384, 420.
DOI URL PMID |
| [13] |
Jurgens G, Lindström K, Saano A (1997) Novel group within kingdom Crenarchaeota from boreal forest soil. Applied and Environmental Microbiology, 63, 803-805.
DOI URL PMID |
| [14] |
Karner MB, DeLong EF, Karl DM (2001) Archaeal dominance in the mesopelagic zone of the Pacific ocean. Nature, 409, 507-510.
DOI URL PMID |
| [15] |
Lueders T, Pommerenke B, Friedrich MW (2004) Stable-isotope probing of microorganisms thriving at thermodynamic limits: syntrophic propionate oxidation in flooded soil. Applied and Environmental Microbiology, 70, 5778-5786.
DOI URL PMID |
| [16] |
Muyzer G, Smalla K (1993) Application of denaturing gradient gel electrophoresis (DGGE) and temperature gradient gel electrophoresis (TGGE) in microbial ecology. Antonie van Leeuwenhoek, 73, 127-141.
URL PMID |
| [17] |
Øvreås L, Forney L, Daae FL, Torsvik V (1997) Distribution of bacterioplankton in meromictic lake Saelenvannet, as determined by denaturing gradient gel electrophoresis of PCR-amplified gene fragments coding for 16S rRNA. Applied and Environmental Microbiology, 63, 3367-3373.
DOI URL PMID |
| [18] |
Pace NR (1997) A molecular view of microbial diversity and the biosphere. Science, 276, 734-740.
DOI URL PMID |
| [19] |
Peng JJ, Lü Z, Rui JP, Lu YH (2008) Dynamics of the methanogenic archaeal community during plant residue decomposition in an anoxic rice field soil. Applied and Environmental Microbiology, 74, 2894-2901.
URL PMID |
| [20] |
Riesner D, Steger G, Zimmat R, Owens RA, Wagenhöfer M, Hillen W, Vollbach S, Henco K (1989) Temperature gradient gel electrophoresis of nucleic acids: analysis of conformational transitions, sequence variations, and protein nucleic acid interactions. Electrophoresis, 10, 377-389.
URL PMID |
| [21] |
Saikaly PE, Stroot PG, Oerther DB (2005) Use of 16S rRNA gene terminal restriction fragment analysis to assess the impact of solids retention time on the bacterial diversity of activated sludge. Applied and Environmental Microbiology, 71, 5814-5822.
DOI URL PMID |
| [22] |
Sandaa RA, Enger TV (1999) Abundance and diversity of archaea in heavy-metal-contaminated soils. Applied and Environmental Microbiology, 65, 3293-3297.
URL PMID |
| [23] | Timonen S, Bomberg M (2009) Archaea in dry soil environments. Phytochemistry Reviews, 8, 505-518. |
| [24] |
van der Wielen P, Voost S, van der Kooij D (2009) Ammonia-oxidizing bacteria and archaea in groundwater treatment and drinking water distribution systems. Applied and Environmental Microbiology, 75, 4687-4695.
URL PMID |
| [25] | Watanabe T, Kimura M, Asakawa S (2006) Community structure of methanogenic archaea in paddy field soil under double cropping (rice-wheat). Soil Biology and Biochemistry, 38, 1264-1274. |
| [26] |
Ye W, Liu X, Lin S, Tan J, Pan J, Li D, Yang H (2009) The vertical distribution of bacterial and archaeal communities in the water and sediment of Lake Taihu. FEMS Microbiology Ecology, 70, 107-120.
DOI URL PMID |
| [27] | Zhu B, Wang T, You X, Gao MR (2008) Nutrient release from weathering of purplish rocks in the Sichuan Basin, China. Pedosphere, 18, 257-264. |
| [1] | Wei Yi, Yi Ai, Meng Wu, Liming Tian, Tserang Donko Mipam. Soil archaeal community responses to different grazing intensities in the alpine meadows of the Qinghai-Tibetan Plateau [J]. Biodiv Sci, 2025, 33(1): 24339-. |
| [2] | Dawei Guan, Li Li, Xin Jiang, Mingchao Ma, Fengming Cao, Baoku Zhou, Jun Li. Influence of long-term fertilization on the community structure and diversity of soybean rhizobia in black soil [J]. Biodiv Sci, 2015, 23(1): 68-78. |
| [3] | Yiyi Long, Liyuan Yang, Wanjin Liao. Identifying cryptic species in pollinating-fig wasps by PCR-RFLP on mtDNA COI gene [J]. Biodiv Sci, 2010, 18(4): 414-419. |
| [4] | Xinxin Sun, Huirong Liu, Fuying Feng, Jianyu Meng, Heng Li, Malina. Diversity and phylogenetic analysis of planktonic bacteria in eutrophic zone of Lake Wuliangsuhai [J]. Biodiv Sci, 2009, 17(5): 490-498. |
| [5] | Lin Xu, Jiajie Xu, Qiaoli Liu, Ruimei Xie, Gehong Wei. Genetic diversity in rhizobia isolated from Sphaerophysa salsula in several regions of northwestern China [J]. Biodiv Sci, 2009, 17(1): 69-75. |
| [6] | Jianping Zhang, Naiyuan Dong, Haobin Yu, Yongjun Zhou, Yongliang Lu, Ruimei Geng, Liuqing Yu. Bacteria diversity in paddy field soil by 16S rDNA-RFLP analysis in Ningxia [J]. Biodiv Sci, 2008, 16(6): 586-592. |
| [7] | Qihui Teng, Hui Cao, Zhongli Cui, Ying Wang, Bo Sun, Hongtao Hao, Shunpeng Li. PCR-RFLP analysis of bacterial 16S rDNA from a typical garden soil in Taihu region [J]. Biodiv Sci, 2006, 14(4): 345-351. |
| [8] | DENG Zhao-Qun, HU Qin-Xue, LIU Sheng, WANG Yao, LIN Mu-Lan. A homologous comparison between mycoplasma and phytoplasma using 16S rDNA PCR amplification and RFLP analysis [J]. Biodiv Sci, 2000, 08(1): 103-105. |
| [9] | FENG Shu-Tang, CHEN You-Chun. Studies on the biological characteristics, ex situ breeding and genetic diversity of Wuzhishan Pig [J]. Biodiv Sci, 1998, 06(3): 172-179. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
Copyright © 2026 Biodiversity Science
Editorial Office of Biodiversity Science, 20 Nanxincun, Xiangshan, Beijing 100093, China
Tel: 010-62836137, 62836665 E-mail: biodiversity@ibcas.ac.cn